Written by Jennifer Huron on March 25, 2021
Washington, DC, March 25, 2021—The National Organization for Rare Disorders (NORD®) today announced the findings of a new study that details the number of orphan products, generics and biosimilars available to treat rare diseases. NORD commissioned Avalere to conduct the analysis to… Read More
Written by Jennifer Huron on March 4, 2021
Washington, DC, March 4, 2021—As US policymakers consider options to address rising health care costs while still meeting the needs of patients, a new report from IQVIA has found that rare diseases account for just 11% of medical invoice spending in the United States, and 79% of all orphan… Read More
Written by Valaree DonFrancesco on November 19, 2020
Washington, DC—November 19, 2020: Today, the National Organization for Rare Disorders (NORD®) published a comprehensive report through its RareInsights™ initiative: Barriers to Rare Disease Diagnosis, Care, and Treatment in the US: A 30-year Comparative Analysis. As part of an ongoing missionRead More
Written by Valaree DonFrancesco on October 15, 2020

A new article on PsychologyToday.com, “People with Rare Diseases Need Better Social Support,” examines a new study that shows which types of support most benefit those with rare diseases. NORD is mentioned in the article and our RareInsights rare disease factsheet (see above) is prominently featured.
#NORDinthenews
Written by Lisa Sencen on November 19, 2018
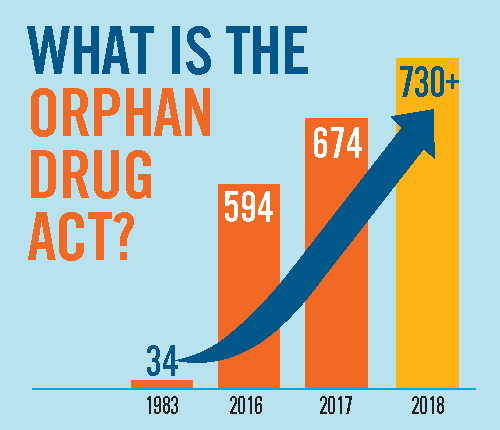
Senator Hatch and Representatives Lance and Butterfield introduced a resolution that heralds the success of the Orphan Drug Act (ODA) and calls for continued support of the legislation.
This year marks the 35th anniversary for the ODA. That means 35 years of increased hope and treatments for the rare disease community…. Read More








